LabConnectA Proven Partner in Your Clinical Research Trial Success
For more than 20 years, LabConnect is a trusted partner helping clients successfully achieve clinical research trial milestones by providing unparalleled program oversight and support.
Services You Can Count On, Across the Globe
Through our innovation and global alliances, we are the world’s most agile central laboratory solutions partner, providing customized, orchestrated, and scalable support services for clinical trials worldwide.
Therapeutic Expertise
With 2000+ clinical research trials across multiple therapeutic areas and disease states, from cell and gene therapy (ATMP) to the latest vaccine candidates and more, LabConnect has the expertise, capability, or model to manage the relevant level of complexity to support clients in successfully achieving their clinical trial milestones.
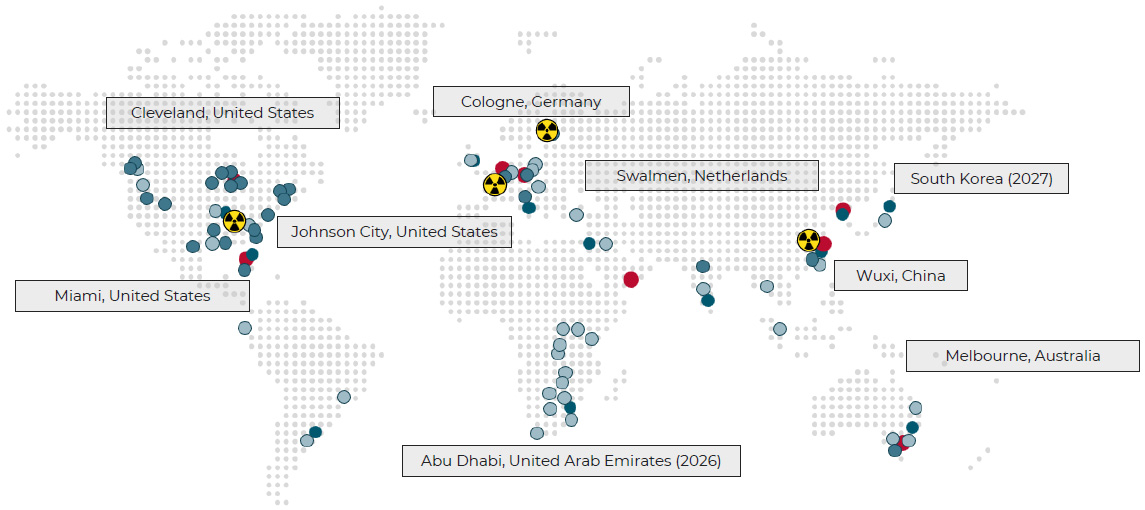
Cell and Gene Therapy
Your cell and gene therapy / ATMP could be the one that changes everything.
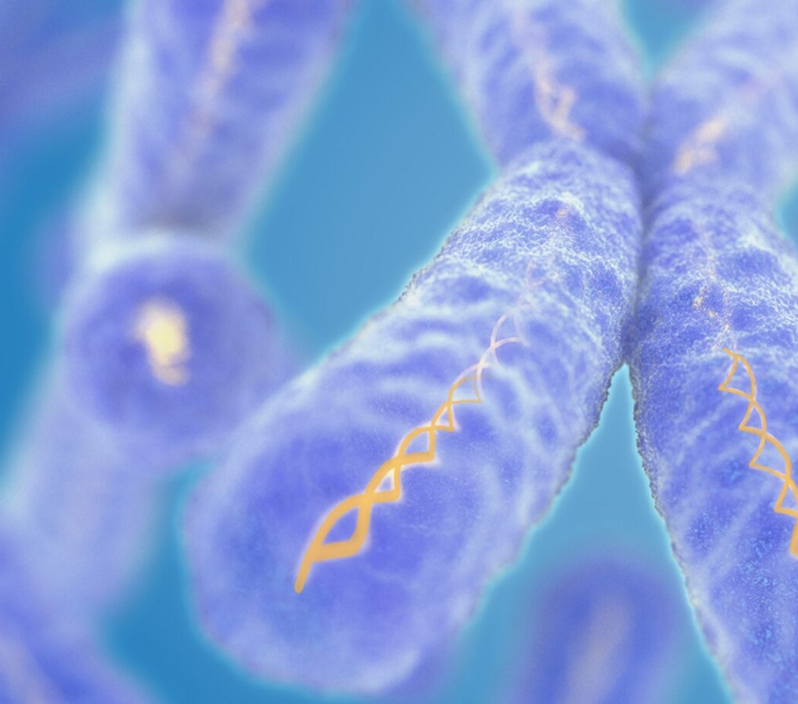
Oncology
Ready to find a cure for your complex challenge? Learn how LabConnect's expertise can accelerate your oncology trial.
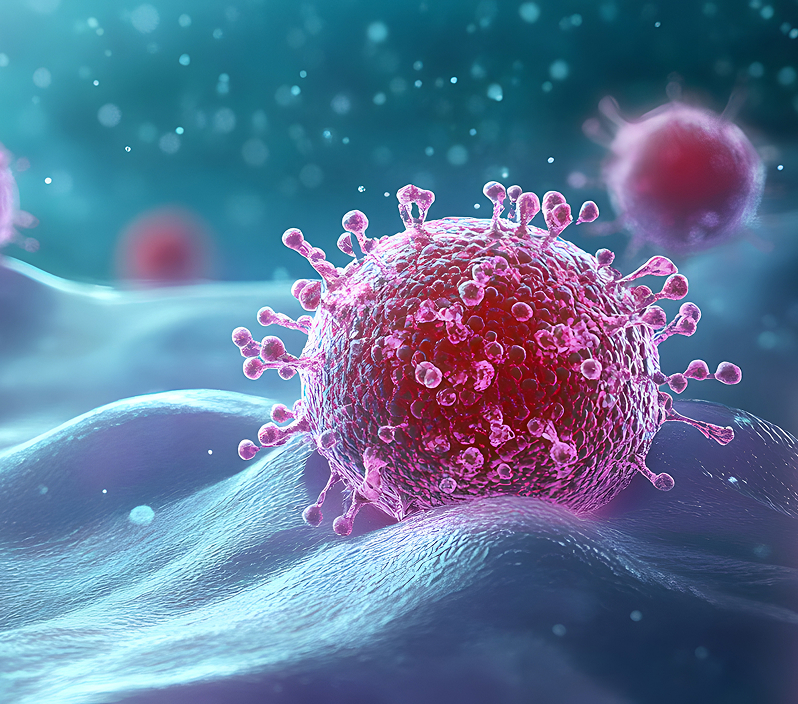
Vaccine
LabConnect is ready to work with you. Together, we can do our part to protect the world from illness and create healthier communities.

And More
In addition to the sample therapeutic areas highlighted above, LabConnect has the expertise, capability, or model to manage the relevant level of complexity across a multitude of therapeutic areas.
Events and Resources
Connect with LabConnect at an upcoming industry conference or congress. Gain insights from our team of experts and explore how we may partner together.

Conferences
June 14-18 DIA Annual 2026 | Philadelphia, PA

News
April 30, 2026LabConnect CEO Announcement: Richard Harris Appointed CEO

Video
March 19, 2025LabConnect’s Global Operations
Contact Us
Explore how we can partner to accelerate the development of new medicines around the world.