Built from Experience Across Millions of SamplesConnected Across the Entire Sample Journey
SampleGISTICS® 2.0 is an application that digitizes and connects clinical trial sample capture at the source, enabling sites to scan, record and transmit participant sample data in real time.
Developed from more than two decades of experience managing samples across global clinical trials, it is designed to integrate site activity directly with laboratories, logistics and operations, making sample data immediately available to support downstream shipment and laboratory processes.
SampleGISTICS 2.0 addresses persistent operational challenges such as paper-based requisitions, manual data entry, and disconnected courier processes that can lead to lost samples, delayed shipments, and limited oversight.
Instead of relying on delayed updates and fragmented systems, SampleGISTICS 2.0 digitizes and connects the workflow at the source, enabling faster, more reliable sample handling from the moment of collection.
Why This Is Only Possible with a Lab Like No Other
SampleGISTICS 2.0 is not a standalone application. It is embedded within LabConnect’s global infrastructure, integrating site workflows directly with laboratories, logistics networks and operational oversight.
Because LabConnect manages the full sample journey, SampleGISTICS® 2.0 can:
- Connect site activity directly to downstream laboratory and logistics processes
- Provide real-time visibility across a global, multi-lab ecosystem
- Align data capture with real-time operational execution, not just documentation
- Eliminate disconnects between collection, shipment and laboratory intake
This level of coordination is only possible through a unified approach to sample management.

Core Capabilities of SampleGISTICS 2.0
Purpose-built for clinical trials and enabled by a lab like no other
Capture at the Source
Barcode-driven scanning at collection ensures precise sample identification
Protocol-Aligned Workflows
Structured, study-specific workflows guide users through collection and data entry
Coordinated Routing, Real-Time Visibility
Directs samples while providing continuous visibility into location, status and movement across each handoff
Built for Global Studies
Supports multi-regional trials with localized language experiences for site users across diverse geographies
Device-Agnostic Access
Browser-based application accessible on any device, with no installs required
Paperless Data Capture
Fully digital workflows eliminate requisitions and manual documentation
Live Study
Sync
Automatically reflects protocol updates, amendments and study changes in real time
Offline Functionality
Enables uninterrupted data capture in low-connectivity environments with automatic sync
From Digital Pen to an Integrated Sample Journey
SampleGISTICS began as a digital pen, improving how participant and visit data was recorded at the site. Over time, LabConnect has supported millions of samples across hundreds of global clinical trials, building deep expertise across laboratories, couriers and operational execution.
As studies have become more complex and decentralized, they have outgrown point solutions.
SampleGISTICS 2.0 was purpose-built for clinical trials, connecting sites, laboratories and logistics into a single, coordinated workflow. It is not a standalone tool, but an extension of LabConnect’s operational approach, integrating sample capture at the site directly into the broader process that supports the full sample journey from collection through laboratory intake.
SampleGISTICS® 2.0 reflects a fundamental shift from point solutions to fully integrated sample management, delivered through a lab like no other.


Improving Sample Management Across TrialsPowered by a Lab Like No Other
When sample capture is digitized at the clinical trial site, the impact extends across the entire study.
Sponsors
- Reduce operational risk by minimizing manual steps, data discrepancies, and fragmented handoffs
- Gain real-time visibility into sample location, status, and stability across the entire journey
- Improve coordination across sites, logistics, and laboratories
- Enable faster, more reliable results through immediate data availability for laboratory processing
- Increase confidence in study outcomes with greater control over sample quality and timelines
CROs
- Improve cross-functional coordination across sites, logistics, and laboratories within a single workflow
- Monitor sample activity in real time with centralized, study-wide oversight
- Reduce delays and manual intervention by minimizing rework and data gaps
- Support consistent execution across sites through protocol-aligned digital workflows
- Strengthen delivery confidence with greater control over sample flow and operational timelines
From Collection to Laboratory IntakeConnected Through a Unified Sample Workflow
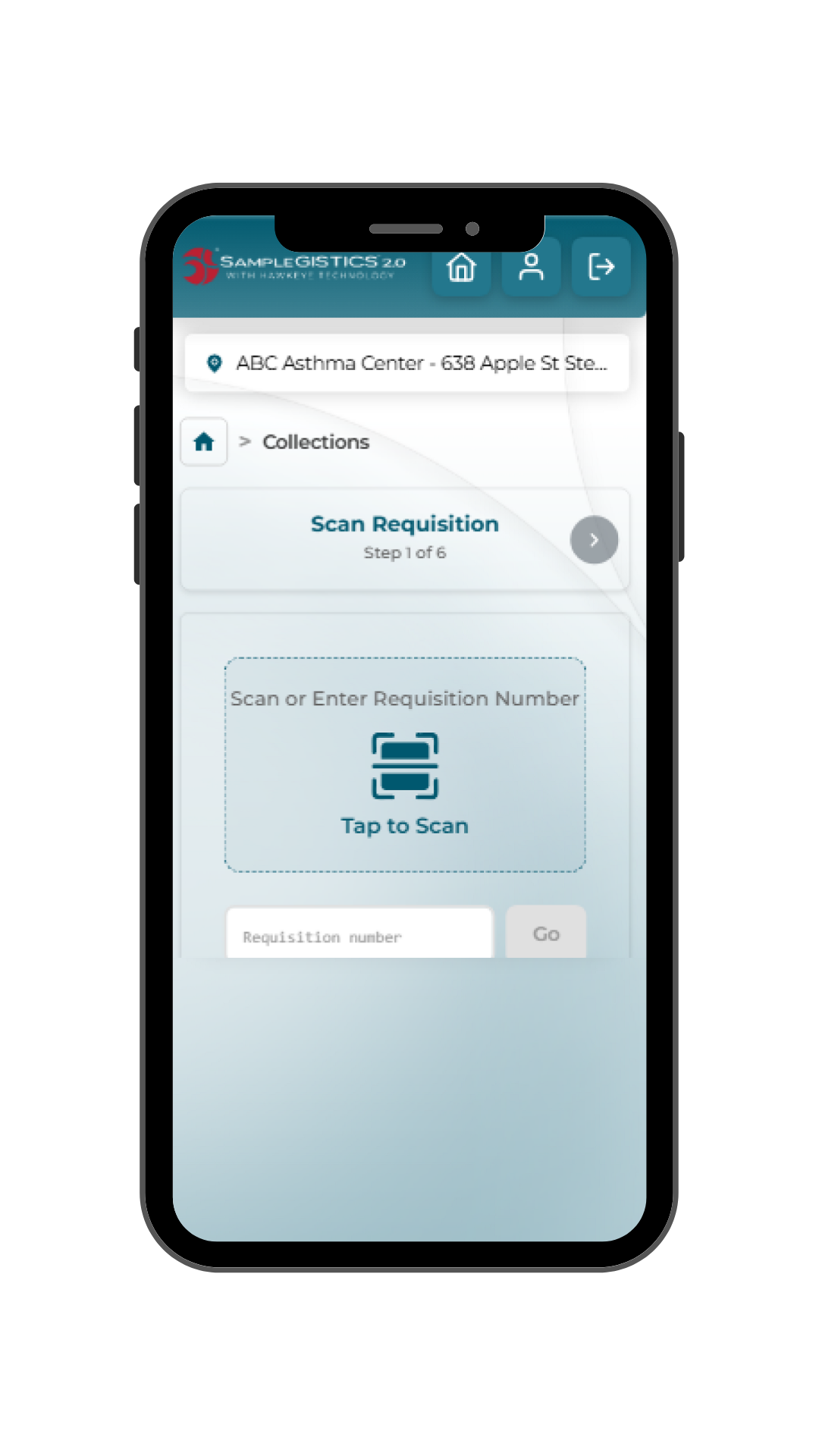
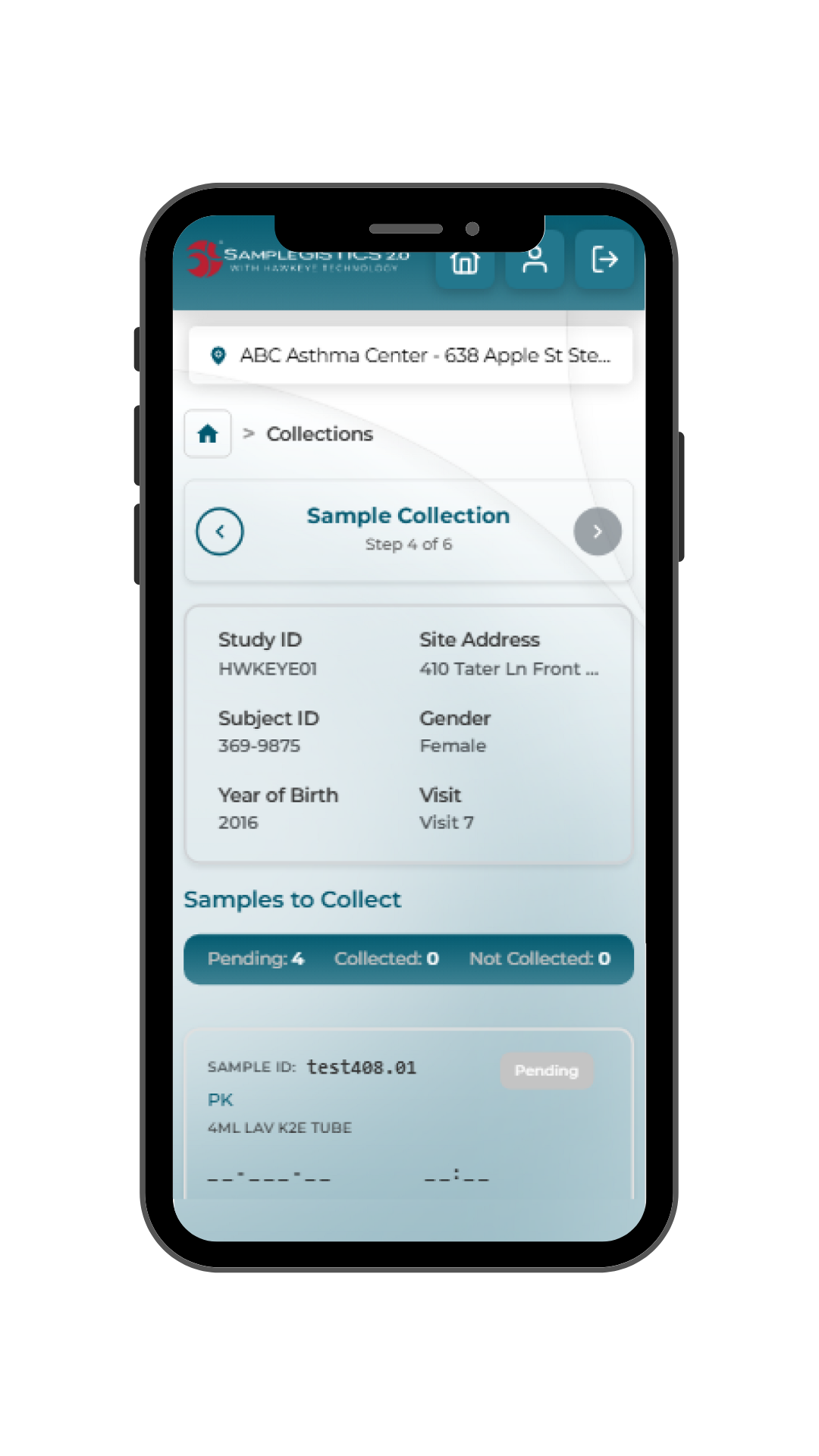
SampleGISTICS 2.0 guides clinical trial site staff through a structured, study-specific workflow that simplifies sample handling while ensuring data is accurate, complete and available.
Each step is digitally connected, ensuring data flows seamlessly from collection through laboratory intake without delays or manual handoffs.
Sample Data Journey
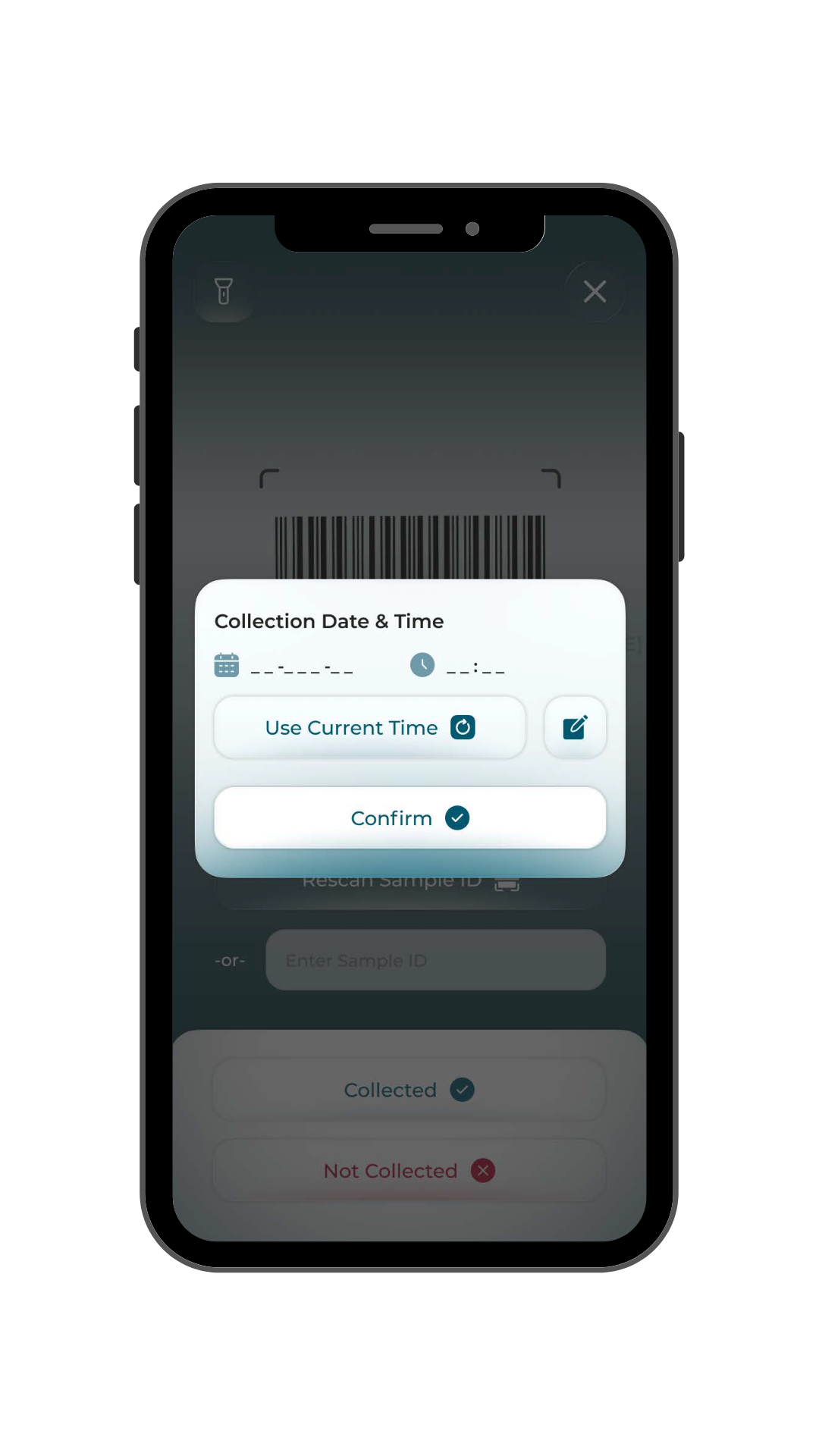
- Collection: Site staff complete requisitions and scan samples directly within the application, reducing manual entry and paperwork.
- Real-time sync: Data is securely transmitted to LabConnect’s validated LIMS environment immediately, eliminating delays and duplicate entry.
- Digitization: Information is automatically structured into standardized data fields, removing the need for manual transcription.
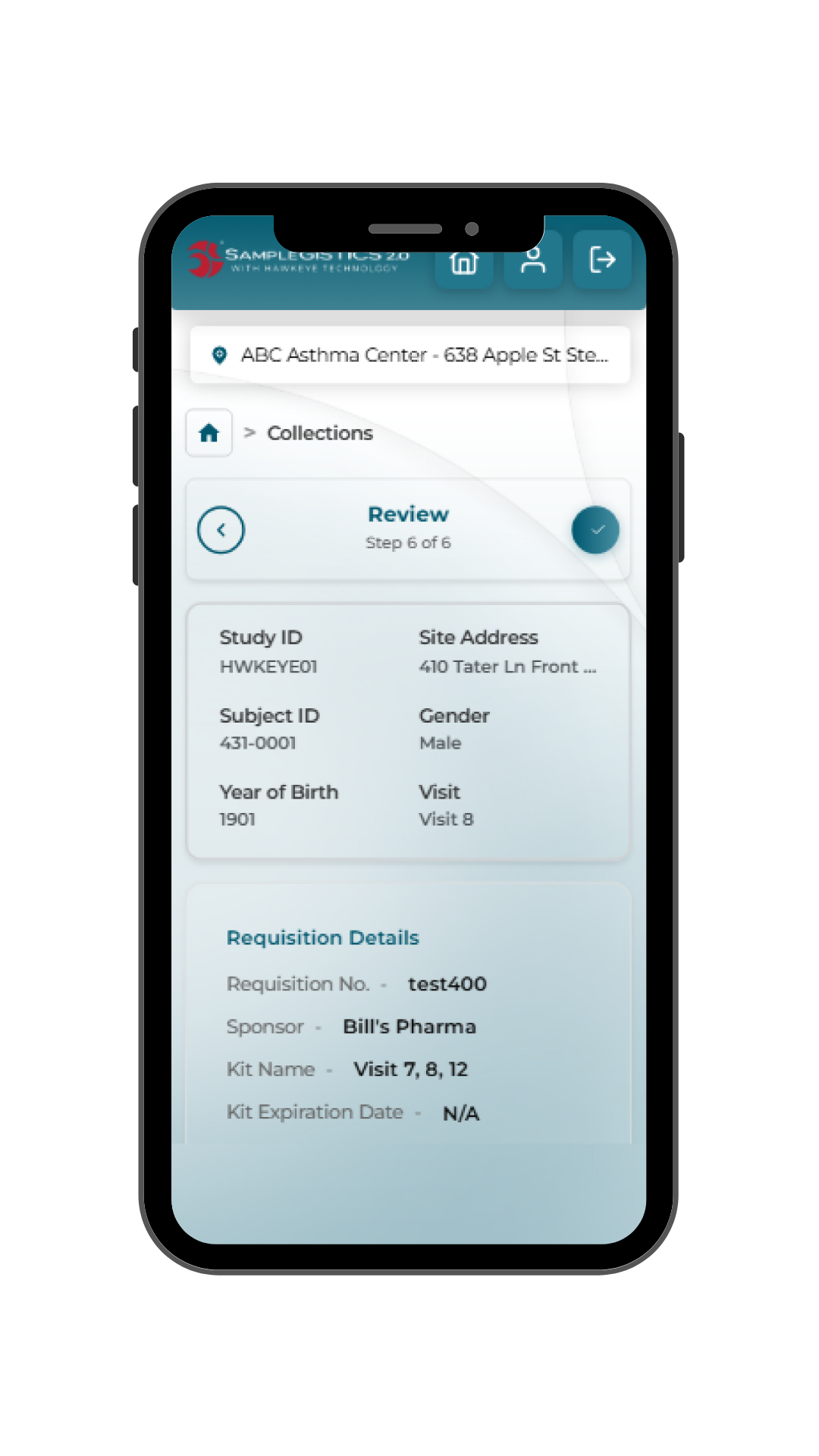
- Validation: Built-in checks confirm completeness, accuracy and protocol alignment before submission.
- Submission: Users review and confirm data with confidence, minimizing queries and rework.
- Downstream readiness: Data is immediately available to support shipment coordination and laboratory processing.
See What’s Only Possible with a Lab Like No Other
Experience how SampleGISTICS® 2.0 connects site workflows to the full sample journey.
Schedule a Meeting