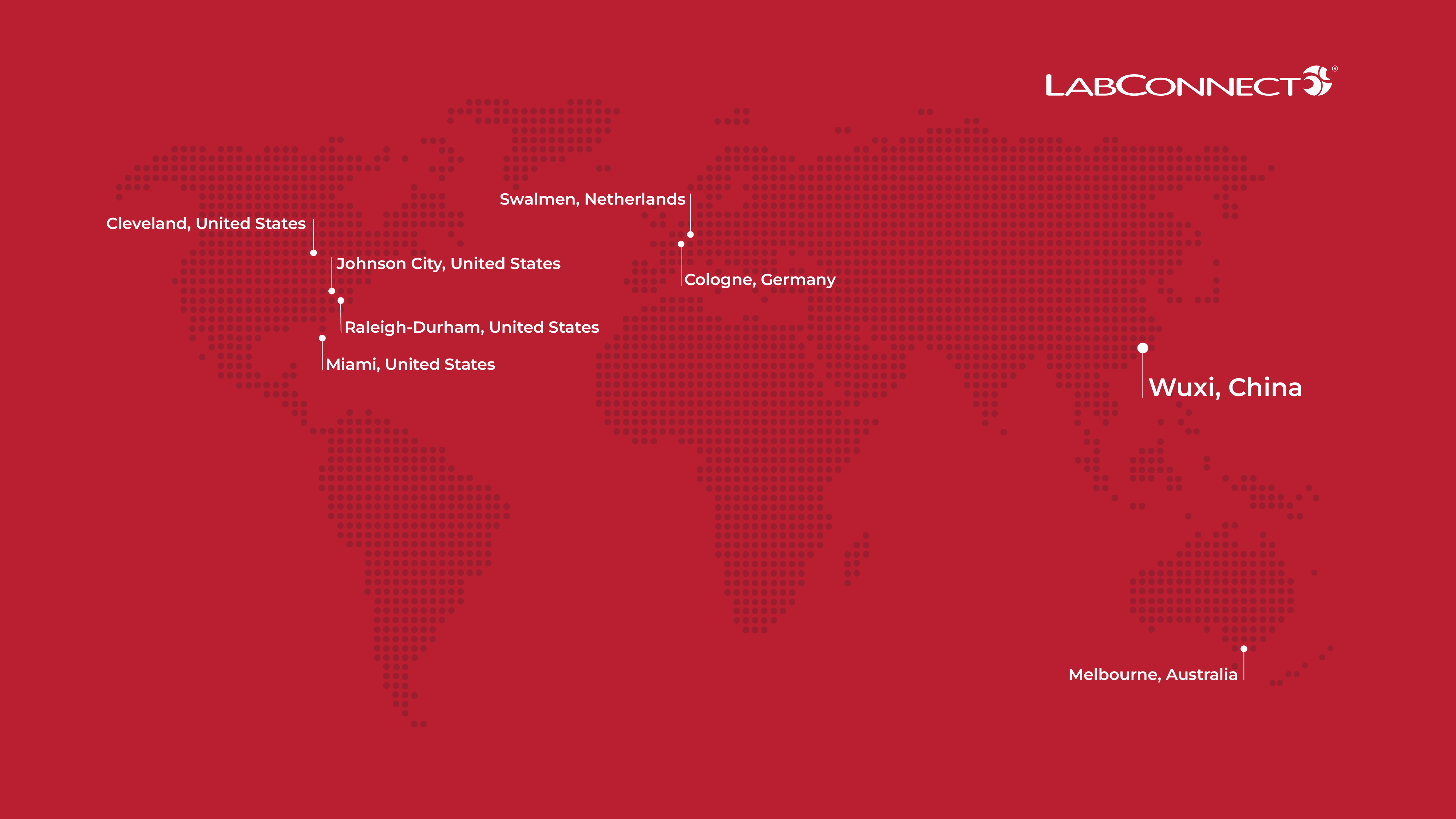
March 11, 2026 — WUXI, China— LabConnect, a leading provider of technology-driven central laboratory services and functional service provider solutions for clinical trials, today announced the grand opening of its new facility in Wuxi, China. As clinical trials become increasingly global and complex, sponsors are seeking laboratory partners that can provide integrated testing, logistics and data oversight across regions. Together with LabConnect's laboratory capabilities in Australia, the new site strengthens the company's ability to support multi-regional clinical trials while helping Asia-Pacific pharmaceutical, biotechnology and clinical research organizations expand their research programs globally.
Fully integrated into LabConnect's global infrastructure, the Wuxi facility enables study teams to operate across regions with unified data, consistent operational oversight and advanced sample logistics and handling procedures.
The facility was developed in collaboration with Teddy Laboratory, now part of Frontage Laboratories, combining Teddy Laboratory's local laboratory expertise with LabConnect's global central laboratory services, logistics capabilities, and technology-driven decentralized network model. "We are proud to offer China-based capabilities and a global service network that supports both multinational pharmaceutical and biotechnology companies entering China and Chinese companies expanding into international markets," said Wes Wheeler, Chief Executive Officer of LabConnect.
The Wuxi facility will support a range of clinical trial services, including custom kit building, advanced sample tracking and logistics, and biorepository services, all designed to meet international regulatory standards.
The grand opening event, held today at the facility in the Xinwu District of Wuxi, included a ceremonial ribbon cutting, guided laboratory tours and presentations highlighting LabConnect's technology-driven approach to central laboratory services. Representatives from LabConnect, Teddy Laboratory, government officials, and members of the regional clinical research community attended the event.
With the addition of the Wuxi site, LabConnect now operates eight global locations, enabling sponsors and CROs to rely on a single partner for central laboratory services across studies of any size or geographic complexity.
For additional information about LabConnect's clinical trial services and laboratory capabilities in China, visit our Chinese-language page.
LabConnect has expanded its global central laboratory network with a new clinical trial services facility in Wuxi, China, strengthening support for multi-regional studies across Asia-Pacific and worldwide.
Key Takeaways
- LabConnect has opened a new central laboratory facility in Wuxi, China, expanding its global network supporting multi-regional clinical trials.
- The facility strengthens LabConnect's ability to support clinical research across Asia-Pacific, alongside its laboratory capabilities in Australia.
- The site provides central laboratory services including clinical trial kit building, sample logistics and tracking, and biorepository support.
- The Wuxi facility is integrated into LabConnect's global network connecting laboratories, logistics, technology and scientific expertise to support complex clinical trials.
- The expansion supports both multinational pharmaceutical companies conducting research in China and Asia-Pacific biotech companies expanding globally.
Why Wuxi, China?
Wuxi has become one of China's leading centers for biotechnology and pharmaceutical development, with a rapidly growing ecosystem of life sciences companies, research organizations and clinical development programs. Its proximity to major pharmaceutical hubs and expanding clinical research infrastructure has helped position the region as an important hub for global drug development.
Asia-Pacific represents one of the fastest growing regions for clinical research and now accounts for roughly a quarter of the global central laboratory market. Regulatory reforms, increasing collaboration in international drug development, and rising clinical trial activity across the region continue to accelerate that growth.
Establishing a presence in Wuxi allows LabConnect to support sponsors conducting research in China while connecting those programs to its broader global laboratory network.
"This facility reflects LabConnect's continued investment in building the global services needed to support clinical trials today and the next generation of clinical research."
— Wes Wheeler, LabConnect Chief Executive Officer
FAQs
Where is LabConnect's new clinical trial laboratory facility located?
LabConnect's newest facility is located in the Xinwu District of Wuxi, China, a major biotechnology and pharmaceutical development hub in the Asia-Pacific region.
What services does the LabConnect Wuxi facility provide?
The facility supports clinical trial services including kit building, sample logistics and tracking, biorepository services and central laboratory support for multi-regional clinical trials.
How does the Wuxi facility support global clinical trials?
The site is integrated into LabConnect's global laboratory network, allowing study teams to manage samples, data and testing across multiple regions with consistent operational oversight.
 We are the world's most agile central laboratory solutions partner.
We are the world's most agile central laboratory solutions partner.
About LabConnect
LabConnect is a global provider of technology-driven central laboratory services and functional service provider solutions for pharmaceutical, biotechnology and contract research organizations. The company connects laboratories, logistics, technology and scientific expertise through a decentralized laboratory network model to deliver integrated solutions for clinical trials worldwide.
Let’s Get Started
Explore how you can accelerate your clinical trial with LabConnect's end-to-end central lab services and functional service provider solutions.










